|
Russian Pharmaceutical Technologies LLC is a research and development company focused on transforming innovative scientific discoveries into novel therapeutic agents.
|
Brief company description
City and country: Saint-Petersburg, Russian Federation Date of foundation: 12 April 2012 Residence status: Skolkovo Foundation Resident, Moscow Team: Russian and World experts in oncology and drug development Brief product description Product: Alofanib, allosteric inhibitor of fibroblast growth factor receptor 2 Indication: FGFR2-expressing tumors Stage of development: Phase 1b clinical study Expected date of completed Phase 1b clinical study: July 2020 |
|
Alofanib (RPT835) is first allosteric FGFR2 inhibitor in oncology
Allosteric inhibitors decrease activity of receptor by binding an allosteric site at the receptor (that is, a site other than the receptor's active site). There are no approved allosteric targeted agents in oncology. Alofanib (RPT835) is a low-molecular weight allosteric inhibitor of FGFR2. Principle benefits of allosteric inhibitors: High specificity | Low toxicity | Cells with mutations sensitive to treatment | Different isoforms of receptor could be targeted | 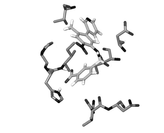
Alofanib binds to allosteric site of extracellular part of FGFR2 and changes the conformation of receptor
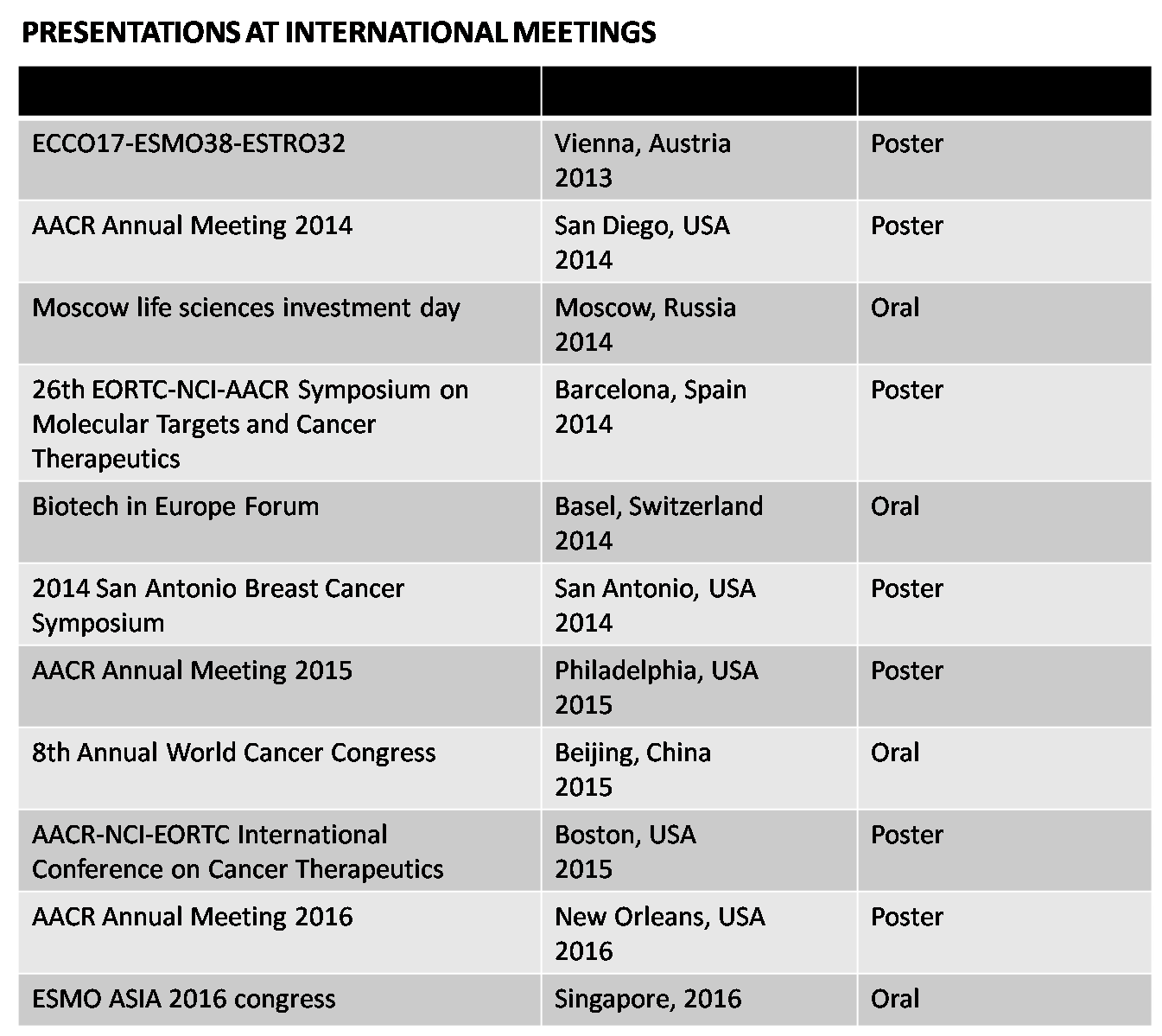
Characterisation and comparison of RPT835
The preclinical data obtained with RPT835 are consistent with potent inhibition of FGF signaling, angiogenesis, and cancer growth. Results were presented at the different international meetings THE FIRST RUSSIAN TARGETED THERAPY DRUG DEVELOPED WITH THE SUPPORT OF SKOLKOVO FOUNDATION HAS ENTERED THE CLINICAL TRIAL STAGE IN ONCOLOGY
The Ministry of Healthcare of the Russian Federation has approved the conduct of the clinical trial to evaluate safety and preliminary efficacy of alofanib which is the first Russian targeted agent in oncology. Alofanib will be studied in gastric cancer patients who have exhausted other standard treatment options. Alofanib was developed by a Russian company “Russian Pharmaceutical Technologies” ("Ruspharmtech", St. Petersburg). The company has created an innovative Russian drug with the support of Skolkovo Foundation. Alofanib is the world’s first drug from a class of allosteric inhibitors of the fibroblast growth factor receptor 2 (FGFR2). Many types of the tumor cells including gastric cancer cells express the FGFR2 receptor on their surface. The receptor activation results in the cell reproduction, tumor growth and metastases. Unlike existing inhibitors that work inside the cell alofanib blocks the part of the receptor that is located outside of the cell, convoluting the receptor which results in its deactivation.
This mechanism allows for affecting the receptor regardless of various FGFR isoforms that may appear (whereas monoclonal antibodies become no longer effective in this situation) and despite new mutations that result in treatment resistance of a tumor cell (whereas tyrosine kinase inhibitors become no longer effective in this situation). This mechanism of action was deemed innovative and was patented by “Ruspharmtech” in Russia, EEU countries, USA, European Union, Japan and China. The results of the preclinical studies published in the recognized international journals, such as European Journal of Cancer, Medicinal Chemistry and Investigational New Drugs, demonstrate the need of further research of the drug in the clinical trials. Currently there is a start of the phase 1b clinical trial in patients with metastatic gastric cancer for whom the standard treatment options proved to be ineffective. The study will be conducted in Moscow, St. Petersburg, Omsk and Rostov-on-Don. "Gastric cancer is an important medical and social problem in Russia, - says Prof. Sergei Tjulandin, Chairman of the Russian Society of Clinical Oncology, Deputy Director of the N.N. Blokhin Russian Cancer Research Center of the Ministry of Health of the Russian Federation. – This type of tumor has been the 4th and 6th most prevalent cancer in men and women, respectively, with over 37,000 new cases ofgastric cancers each year. Unfortunately once metastases appear our treatment options become very limited. Life expectancy of such patients is about 1 year". “The Skolkovo Foundation has been actively supporting the development of targeted therapy that affect a specific target within tumors”, – says senior vice-president for innovations in Skolkovo Foundation, Kirill Kaem. – "New types of inhibitors can result in dramatic changes and affect the patient's life expectancy, which is already happening in case of lung cancer, breast cancer, kidney cancer and other tumors. The first Russian targeted drug has successfully passed a number of extended preclinical trials in accordance with international requirements and will enter the first stage of the clinical trials to be studied in gastric cancer patients. The Grants Committee of the Foundation made a decision on further support of our project". |
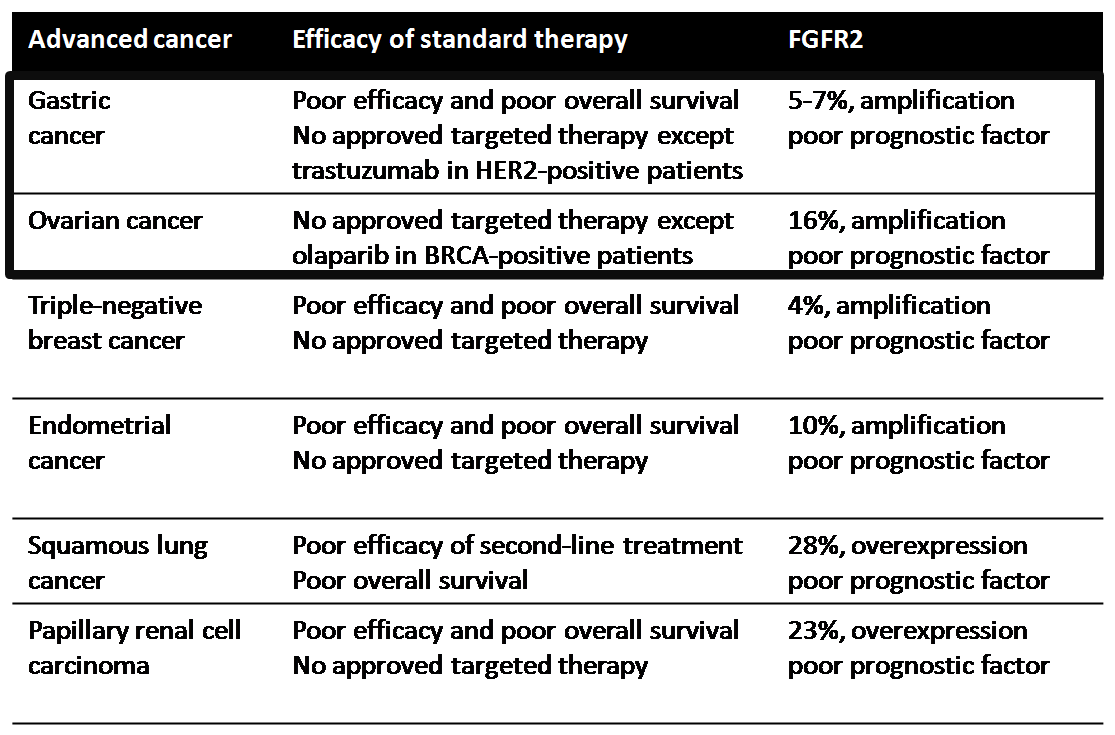
Fibroblast Growth Factor Receptor 2 as a target for RPT835
The recent identification of fibroblast growth factor receptor 2 (FGFR2) overexpression or mutations in different cancer types has generated an opportunity for a novel target-based therapy. Publications

ESMO Asia 2016: First-in-class inhibitor of FGFR2 shows promising results in preclinical studies SINGAPORE – A novel allosteric fibroblast growth factor receptor 2 (FGFR2) inhibitor, alofanib, has potent anti-tumour and antiangiogenic activity, according to the results of preclinical studies. The results were presented at the ESMO Asia 2016 Congress in Singapore on December 16-19, 2016 (Abstract 151O). The data provided a strong rationale for the evaluation of the compound in patients with FGFR2-expressing cancers. “Alofanib is a novel first-in-class allosteric small-molecular inhibitor of FGFR2. The compound binds to the extracellular domain of FGFR2 and has an inhibitory effect on FGF2-induced phoshphorylation,” said lead author Ilya Tsimafeyeu, MD, PhD, director of the Kidney Cancer Research Bureau and scientific adviser of Ruspharmtech LLC (Russia). “The in vivo data are consistent with alofanib being a predominantly FGFR-selective inhibitor. In preclinical xenograft models, alofanib significantly inhibited aggressive growth of FGFR2 high-expressing triple-negative breast cancer and had moderate activity in the FGFR2 low-expressing triple-negative breast cancer. Treatment with alofanib did not result in FGFR2-negative lung cancer growth. Accordingly, we believe that alofanib is capable of clinically testing an FGFR tumour-driven hypothesis, aiming to select patients based on deregulated tumour FGFR2 expression,” said Tsimafeyeu. “There is compelling evidence for deregulated FGF/FGFR2 signalling in the pathogenesis of many cancers that originate from different tissue types, for example, gastric and endometrial cancers. This study provides the first pharmacologic profile of the allosteric inhibitor alofanib with potential as a targeted antitumour therapy,” said Olivier Rixe, MD, PhD, professor of the division of hematology/oncology and associate director for clinical research, The Dana Wood endowed chair in cancer therapeutics and early phase clinical research, University of New Mexico Comprehensive Cancer Center (U.S.) Study details In the present study, authors showed that alofanib inhibited phosphorylation of FRS2α with IC50 values of 7 and 9 nmol/L in cancer cells expressing different FGFR2 isoforms (IIIc and IIIb). In a panel of cell lines representing several tumour types (triple negative breast cancer, melanoma, ovarian cancer), alofanib inhibited FGF-mediated proliferation. Alofanib dose dependently inhibited the proliferation and migration of human and mouse endothelial cells (GI50 11-58 nmol/L) compared with brivanib and bevacizumab. Treatment with alofanib ablated experimental FGF-induced angiogenesis in vivo. In a FGFR-driven human tumour xenograft model, oral administration of alofanib was well tolerated and resulted in potent anti-tumour activity. Importantly, alofanib was effective in FGFR2-expressing models. For example, alofanib significantly inhibited aggressive growth of triple-negative breast cancer tumour xenograft (SUM 52PE). The tumour growth curve shows a nearly exponential increase in the median tumour volume up to day 31 in the vehicle group and a high rate of slow-growing tumours up to day 40 in the alofanib group. Early data suggest that combining FGFR2 inhibitors with platinum-containing cytotoxic agents for the treatment of epithelial ovarian cancer may yield increased anti-tumour activity. FGFR2 amplification and high-level polysomy may be a promising molecular target for serous ovarian cancer. FGFR2 amplification was found in 17.1 percent of patients with advanced serous ovarian cancer. Thus, the aim of the presented xenograft studies was to increase the efficacy of chemotherapy by adding the alofanib FGFR2 inhibitor. As expected, intravenous alofanib significantly potentiated the efficiency of the combination of paclitaxel and carboplatin. A daily intravenous regimen resulted in a three-fold tumour growth delay in comparison with the vehicle, and an inhibition rate of 80 percent was achieved. The proliferative index Ki-67 was significantly reduced in mice treated with alofanib. In this study, antiangiogenic activity in an ovarian carcinoma model using a combination of alofanib and chemotherapy was observed. Alofanib decreased the number of vessels by 49 percent and initiated vascular changes in the tumours. “In acute and chronic toxicity studies, alofanib was well-tolerated,” said Dr. Tsimafeyeu. “There was no treatment-related mortality, severe toxicity or significant changes to the organs in the six-month study. Body weight remained stable and it was comparable to vehicle groups. We found new side effects such as the suppression of spermatogenesis. Spermatocytes express FGFR2, and theoretically alofanib could inhibit the proliferation of these cells,” he said. “A phase Ib clinical study protocol has been selected for the ECCO-AACR-EORTC-ESMO Workshop on Methods in Clinical Cancer Research, better known as the ‘Flims’ Workshop, and a clinical study will be initiated at the beginning of 2017,” said Dr. Tsimafeyeu. The studies were supported by a grant from the Skolkovo Foundation. |